Structure-guided Antibiotics
Andrés Palencia
National Institute of Health and Medical Reserves (INSERM)
Published March 27, 2026
In pathogens, RNA carries genetic instructions that are responsible for some of the world’s deadliest and intractable infections.
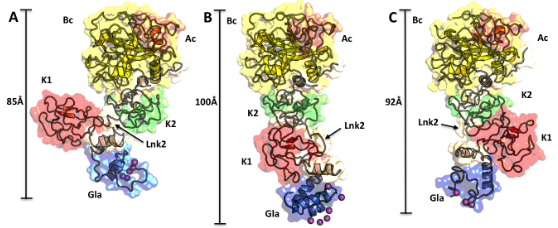
By targeting RNA in the iconic genetic flow from DNA to RNA to protein in pathogens, researchers in the structural biology laboratory of Andrés Palencia are helping to advance new classes of drugs to combat drug-resistant bacteria and parasites that cause diseases ranging from malaria to the severe diarrheal disease. And one promising new inhibitory molecule against tuberculosis that emerged from the work, ganfeborole, has completed a phase 2 clinical trial.
“We have a strong focus in trying …
Find out More »